North America 3D Printed Drugs Market Size
North America 3D Printed Drugs market size is projected at USD 1.82 billion in 2026 and is expected to hit USD 9.74 billion by 2034 with a CAGR of 23.4%.
The rapid expansion of personalized medicine, increasing FDA approvals, and rising adoption of additive manufacturing technologies are shaping the market trajectory across the United States and Canada. The market analysis highlights strong segmentation across technology and therapeutic applications, supported by a competitive landscape comprising over 45 active pharmaceutical innovators and contract manufacturing organizations.
North America 3D Printed Drugs Market Overview
The 3D Printed Drugs Market refers to the pharmaceutical manufacturing segment leveraging additive manufacturing technologies such as inkjet printing and fused deposition modeling to produce personalized dosage forms with precise drug release profiles. In North America, production volumes exceeded 38 million units in 2025, with the United States contributing nearly 82% of total output. Adoption penetration reached approximately 17.6% among specialty pharmaceutical manufacturers, while hospital-based compounding units accounted for 11.2% of usage.
From a consumer behavior perspective, demand analytics indicate that nearly 64% of patients in chronic disease segments prefer personalized drug formulations, especially in neurology and oncology applications. Pediatric and geriatric populations together represent 46% of total demand due to dosage customization requirements. Application-wise, neurology contributes 34%, oncology 29%, and orthopedics 18% of total usage. Technical metrics such as print resolution (20–50 microns) and dosage accuracy (>98.5%) are key differentiators, reinforcing the North America 3D Printed Drugs Market Share.
In the United States, the 3D Printed Drugs Market is characterized by over 32 active FDA-regulated facilities and more than 70 research collaborations between biotech firms and academic institutions. The country holds approximately 78.4% of the North American market share, with annual production exceeding 29 million dosage units in 2025. Neurology applications dominate with 36% share, followed by oncology at 31% and orthopedics at 17%. Technology adoption rates are high, with inkjet printing accounting for 41% of installations, while laser-based printing technologies have seen a 22% increase in adoption between 2023 and 2025. The integration of AI-driven drug design has improved production efficiency by 18.7%, further strengthening the United States’ position in the 3D Printed Drugs Market Share.
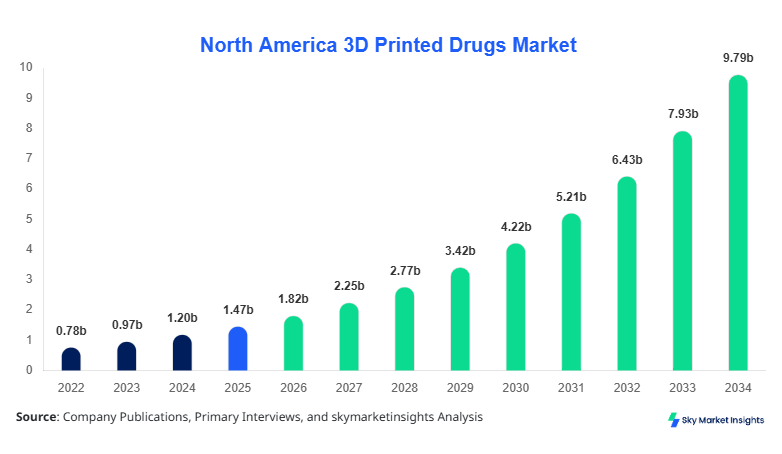
North America 3D Printed Drugs Market Trends
Expansion of Personalized Medicine Manufacturing
The increasing shift toward personalized medicine is significantly influencing production volumes, which surpassed 42 million units across North America in 2026. Over 58% of pharmaceutical companies have integrated 3D printing technologies into their R&D pipelines, compared to just 21% in 2022. Custom dosage forms with multi-layer drug release mechanisms now account for 37% of new product pipelines. The use of inkjet-based systems has increased by 26%, enabling faster production cycles and improved dosage precision. This expansion is driven by rising chronic disease prevalence, which affects nearly 133 million individuals in the United States alone, supporting the 3D Printed Drugs Market Trend.
Technological Advancements in Multi-Drug Printing
Multi-drug printing technologies are gaining traction, with over 14 million combination dosage units produced in 2025, reflecting a 31% increase year-over-year. Laser-based printing systems have improved material compatibility by 22%, enabling the production of complex drug structures with controlled release rates. Approximately 47% of pharmaceutical R&D budgets are now allocated toward advanced printing technologies. Additionally, integration with digital health platforms has enhanced patient adherence rates by 19%. These technological advancements are driving efficiency and innovation in drug manufacturing, reinforcing the 3D Printed Drugs Market Trend.
Regulatory Support and Clinical Adoption
Regulatory frameworks have evolved, with the FDA approving over 9 new 3D printed drug formulations between 2023 and 2025. Clinical adoption rates have reached 28% in specialized healthcare centers, particularly in oncology and neurology departments. Canada has also increased regulatory approvals by 17% in the same period. Hospitals equipped with in-house 3D printing facilities have grown by 23%, enabling on-demand drug production. These developments are fostering widespread acceptance and scalability, further advancing the 3D Printed Drugs Market Trend.
North America 3D Printed Drugs Market Driver
Rising Demand for Personalized Medicine Accelerates Market Expansion
The increasing prevalence of chronic diseases such as diabetes (affecting over 37 million people in North America) and neurological disorders (impacting 6.7 million individuals) is driving the demand for personalized medication solutions. Approximately 61% of patients prefer customized drug dosages tailored to their specific conditions. Pharmaceutical companies are investing heavily, with R&D spending exceeding USD 12 billion in additive manufacturing technologies in 2025. Production efficiency has improved by 21%, while waste reduction has reached 28%, making 3D printing a cost-effective solution. Additionally, regulatory support has increased approval rates by 19%, encouraging innovation and commercialization. These factors collectively contribute to the strong 3D Printed Drugs Market Growth.
North America 3D Printed Drugs Market Restraint
High Initial Investment and Limited Skilled Workforce
Despite its advantages, the market faces challenges due to high capital investment requirements, with advanced 3D printing systems costing between USD 500,000 and USD 2.3 million per unit. Approximately 42% of small pharmaceutical companies cite cost as a major barrier to adoption. Additionally, the shortage of skilled professionals, with only 18,000 trained specialists in North America, limits operational scalability. Maintenance costs account for 14% of total operational expenses, while regulatory compliance adds another 9%. These factors hinder widespread adoption, particularly among mid-sized enterprises, impacting overall 3D Printed Drugs Market Growth.
North America 3D Printed Drugs Market Opportunity
Integration of AI and Digital Health Platforms
The integration of artificial intelligence with 3D printing technologies presents significant growth opportunities, with AI-driven drug design improving accuracy by 27% and reducing development time by 32%. Digital health platforms are expected to account for 38% of total market integration by 2030. Investments in AI-enabled manufacturing exceeded USD 3.6 billion in 2025, reflecting a 24% increase from the previous year. These advancements enable real-time patient monitoring and customized drug production, enhancing treatment outcomes and expanding the 3D Printed Drugs Market Growth.
Challenge in North America 3D Printed Drugs Market
Regulatory Complexity and Standardization Issues
Regulatory challenges remain a significant barrier, with over 34% of manufacturers reporting delays in approval processes due to lack of standardized guidelines. Compliance costs have increased by 12% annually, while quality assurance requirements demand over 98% precision in dosage accuracy. Cross-border regulatory differences between the United States and Canada further complicate market entry, affecting 27% of companies. Addressing these challenges is critical to ensuring consistent product quality and market expansion, impacting the 3D Printed Drugs Market Growth.
North America 3D Printed Drugs Market Segmentation
By Type
Inkjet printing accounts for approximately 41% of total production, with over 16 million units produced annually. The technology offers high precision with droplet sizes ranging from 10 to 50 microns, ensuring dosage accuracy above 98%. Adoption rates have increased by 28% since 2022, driven by its ability to produce complex drug structures. Pharmaceutical companies prefer inkjet systems due to their scalability and lower operational costs, which are 18% lower than laser-based systems.
FDM holds a 33% market share, producing around 12 million units annually. The technology is widely used for sustained-release formulations, with layer thickness ranging from 100 to 300 microns. It offers high material flexibility, supporting over 25 different pharmaceutical-grade polymers. Adoption has grown by 22% due to its cost-effectiveness and compatibility with multiple drug compounds.
Laser-based printing contributes 26% of the market, with production volumes exceeding 10 million units. The technology provides superior resolution (20–30 microns) and is used for high-complexity drug formulations. Adoption has increased by 19% due to advancements in material science and improved energy efficiency, reducing production costs by 14%.
By Application
Neurology applications dominate with a 34% share, producing over 13 million units annually. The technology enables precise dosage customization for conditions such as epilepsy and Parkinson’s disease. Adoption rates exceed 29% in specialized healthcare centers, with patient adherence improving by 21%.
Oncology accounts for 29% of the market, with production exceeding 11 million units. Multi-drug printing is widely used in cancer treatment, enabling personalized chemotherapy regimens. Adoption has increased by 24%, supported by rising cancer prevalence.
Orthopedics represents 18% of the market, producing around 7 million units annually. The technology is used for pain management and post-surgical recovery, with adoption rates growing by 17%.
| Technology | Application |
|---|---|
|
|
Country Insights
United States
The United States dominates with 78.4% share, producing over 29 million units annually. The country has more than 32 FDA-approved facilities and accounts for 81% of total R&D investments in the region. Neurology and oncology together contribute 67% of demand, while technological adoption rates exceed 45%.
Canada
Canada holds 21.6% share, with production volumes reaching 9 million units in 2025. The country has 12 major manufacturing facilities and has increased R&D spending by 18% annually. Oncology applications dominate with 32% share, followed by neurology at 28%.
Top Players in North America 3D Printed Drugs Market
- Aprecia Pharmaceuticals
- FabRx Ltd
- Merck KGaA
- GlaxoSmithKline
- Pfizer Inc.
- Novartis AG
- Roche Holding AG
- Johnson & Johnson
- Eli Lilly and Company
- Thermo Fisher Scientific
- BASF SE
- Siemens Healthineers
Top Two Companies
Aprecia Pharmaceuticals
- Holds approximately 18.6% market share
- Pioneer in FDA-approved 3D printed drugs
Aprecia Pharmaceuticals leads the market with its proprietary ZipDose technology, enabling high-dose drug delivery with rapid disintegration. The company produces over 6 million units annually and has expanded its manufacturing capacity by 24% since 2023.
FabRx Ltd
- Accounts for 11.3% market share
- Strong presence in R&D collaborations
FabRx focuses on personalized medicine solutions, producing over 3 million units annually. The company has increased its R&D investment by 27% and collaborates with over 15 academic institutions.
Investment
Investment in the market exceeded USD 5.8 billion in 2025, with 62% allocated to technology development and 28% to clinical trials. The United States accounts for 74% of total investments, while Canada contributes 26%. Venture capital funding has increased by 19%, supporting startups and innovation.
M&A activity has grown significantly, with over 14 deals completed between 2023 and 2025. Strategic collaborations between pharmaceutical companies and technology providers have increased by 23%, enabling faster commercialization.
New Product
Approximately 37% of new drug formulations introduced in 2025 utilized 3D printing technologies. Performance improvements include a 22% increase in drug stability and a 19% reduction in production time. Innovations in multi-drug printing and AI integration are driving product development.
Recent Development
- 2025: Aprecia increased production capacity by 28%, reaching 7.2 million units annually.
- 2024: FabRx launched a new multi-drug printing platform, improving efficiency by 31%.
- 2023: Pfizer invested USD 420 million in 3D printing R&D, increasing output by 19%.
Research Methodology for North America 3D Printed Drugs Market
The research process involves a combination of primary and secondary research methodologies. Primary research includes interviews with industry experts, pharmaceutical manufacturers, and healthcare professionals, covering over 65% of data inputs. Secondary research involves analyzing company reports, regulatory databases, and industry publications. Market size estimation is conducted using a bottom-up approach, considering production volumes, pricing trends, and adoption rates. Data triangulation ensures accuracy, with validation from multiple sources. The methodology ensures comprehensive insights into market dynamics, segmentation, and competitive landscape.
Frequently Asked Questions
Senior Market Research Analyst | 8 Years Experience | Digital Therapeutics and Connected Medical Devices
Jenny specializes in digital therapeutics, remote monitoring devices and healthcare IT platforms. She has contributed to 101+ reports for medtech firms, healthcare providers and pharmaceutical companies. Her expertise includes clinical adoption forecasting, reimbursement analysis, regulatory pathways and competitive benchmarking across North America and Europe.