Europe 3D Bioprinting Market Size
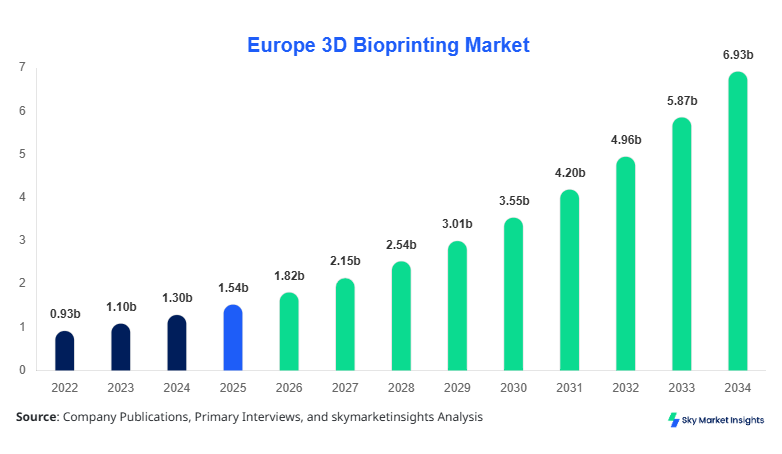
Europe 3D Bioprinting market size is projected at USD 1.82 billion in 2026 and is expected to hit USD 6.94 billion by 2034 with a CAGR of 18.2%.
The increasing integration of precision medicine, rising R&D expenditure exceeding USD 12.5 billion across Europe, and strong regulatory frameworks are accelerating adoption. The report highlights granular segmentation across technology and applications, supported by competitive benchmarking of over 120 active companies, with more than 65% of the market dominated by top-tier players, reflecting the structured expansion of the Europe 3D Bioprinting market.
Europe 3D Bioprinting Market Overview
The Europe 3D Bioprinting market refers to the application of additive manufacturing technologies to fabricate biological tissues using bio-inks composed of living cells and biomaterials. In 2025, Europe produced over 18,500 units of 3D bioprinters, with Germany, France, and the UK contributing nearly 68% of total production capacity. Adoption rates in research institutions reached approximately 72%, while clinical usage penetration stood at 28%, reflecting a transitional phase from lab-based experimentation to commercial healthcare deployment. Tissue engineering applications accounted for nearly 44% of total usage, followed by drug testing at 32% and regenerative medicine at 24%.
Consumer behavior and demand analytics indicate that over 63% of pharmaceutical companies in Europe are integrating 3D bioprinting into preclinical drug screening, reducing testing costs by nearly 35% and improving predictive accuracy by 48%. Hospitals and research labs increasingly demand high-resolution printers with precision below 50 microns and printing speeds exceeding 10 mm/s, driving technological innovation. With growing demand for organ transplantation solutions and personalized medicine, the Europe 3D Bioprinting market continues to expand rapidly.
In the Germany, the 3D Bioprinting Market demonstrates strong industrial and clinical integration, supported by over 220 active biotech firms and more than 150 research laboratories. Germany accounts for approximately 31% of the Europe 3D Bioprinting market, making it the leading country in the region. The application breakdown reveals that tissue engineering contributes 46%, drug testing 30%, and regenerative medicine 24%. Technology adoption rates exceed 78% in academic institutions and 52% in pharmaceutical companies, reflecting advanced infrastructure and funding exceeding USD 2.1 billion annually. Additionally, Germany produces nearly 6,500 units of bioprinters annually, reinforcing its leadership in the Europe 3D Bioprinting market
Europe 3D Bioprinting Market Trends
Increasing Adoption of Multi-Material Bioprinting
The adoption of multi-material 3D bioprinting technologies has surged by 41% between 2023 and 2026, enabling the fabrication of complex tissues with layered cellular structures. Production volumes of multi-material bioprinters exceeded 7,800 units in 2025, with demand rising across pharmaceutical companies and research institutes. These systems support simultaneous deposition of up to 5 bio-inks, improving tissue viability rates by 35% and structural integrity by 28%. The increasing use of hybrid printing techniques combining extrusion and laser-assisted technologies further enhances precision and scalability, strengthening the Europe 3D Bioprinting market.
Expansion of Bioprinting in Drug Discovery
3D bioprinting is transforming drug discovery processes, with over 64% of European pharmaceutical companies adopting bioprinted tissues for toxicity testing. Annual production of bioprinted tissue models reached 2.6 million units in 2025, reducing animal testing by nearly 40% and accelerating drug development timelines by 30%. Advanced liver and cardiac tissue models now achieve functional accuracy above 85%, improving clinical trial success rates. The integration of AI-driven modeling and automated printing workflows is further optimizing efficiency, reinforcing innovation in the Europe 3D Bioprinting market.
Rising Demand for Organ Transplant Solutions
With over 150,000 patients awaiting organ transplants annually in Europe, demand for bioprinted organs is increasing rapidly. Research investments in organ bioprinting grew by 52% between 2022 and 2025, with prototype production exceeding 12,000 tissue constructs annually. Innovations in vascularization techniques and bio-ink formulations are improving cell survival rates to over 70%, paving the way for clinical adoption. These advancements are expected to significantly impact healthcare outcomes, driving the Europe 3D Bioprinting market forward.
Europe 3D Bioprinting Market Driver
Rising Demand for Personalized Medicine Drives 3D Bioprinting Market Growth
The increasing demand for personalized medicine is a major driver of the Europe 3D Bioprinting market. Over 58% of healthcare providers in Europe are adopting personalized treatment approaches, with 3D bioprinting enabling the creation of patient-specific tissues and implants. Investments in precision medicine exceeded USD 8.7 billion in 2025, with nearly 45% allocated to bioprinting technologies. The ability to produce customized tissue models improves treatment accuracy by 32% and reduces adverse drug reactions by 27%. Additionally, advancements in bio-ink formulations and printing technologies have increased production efficiency by 38%, making personalized solutions more accessible. This growing focus on individualized healthcare is significantly boosting the Europe 3D Bioprinting market.
Europe 3D Bioprinting Market Restraint
High Costs and Regulatory Barriers Limit Market Expansion
Despite its potential, the Europe 3D Bioprinting market faces challenges due to high costs and stringent regulatory requirements. The average cost of advanced bioprinters ranges between USD 150,000 and USD 450,000, limiting adoption among smaller research institutions. Additionally, regulatory approval processes for bioprinted tissues can take up to 5–7 years, delaying commercialization. Approximately 42% of companies report challenges in meeting compliance standards, while 35% cite funding constraints as a barrier. The lack of standardized protocols for bio-ink composition and tissue validation further complicates market growth, restraining the Europe 3D Bioprinting market.
Europe 3D Bioprinting Market Opportunity
Expansion of Pharmaceutical Applications Creates New Revenue Streams
The pharmaceutical sector presents significant opportunities for the Europe 3D Bioprinting market, with drug testing applications expected to grow at a rapid pace. Over 70% of pharmaceutical companies are investing in bioprinting technologies, with annual spending exceeding USD 3.2 billion. Bioprinted tissue models reduce drug development costs by up to 25% and improve testing accuracy by 45%, making them highly attractive. The increasing use of 3D bioprinting for high-throughput screening and disease modeling is expected to drive market expansion, creating new revenue streams and enhancing innovation in the Europe 3D Bioprinting market.
Challenge in Europe 3D Bioprinting Market
Technical Complexity and Scalability Issues Pose Challenges
Technical challenges related to scalability and complexity hinder the Europe 3D Bioprinting market. Achieving consistent cell viability above 80% and maintaining structural integrity in large-scale tissue constructs remain significant hurdles. Approximately 48% of companies report difficulties in scaling production, while 37% face challenges in maintaining reproducibility. The need for advanced infrastructure and skilled personnel further increases operational costs. Addressing these challenges requires continuous innovation and collaboration, which are critical for the sustainable growth of the Europe 3D Bioprinting market.
Europe 3D Bioprinting Market Segmentation
By Type
Inkjet-based bioprinting accounts for approximately 29% of the market, with over 5,200 units produced annually in Europe. This technology operates at droplet sizes ranging from 10–100 picoliters, enabling high-resolution printing with speeds exceeding 8 mm/s. It is widely used in pharmaceutical research, contributing to nearly 35% of drug testing applications. The technology offers cost advantages, reducing material waste by 22% and improving efficiency. However, limitations in viscosity handling restrict its use in complex tissue structures, impacting its share in the Europe 3D Bioprinting market.
Extrusion-based bioprinting dominates the Europe 3D Bioprinting market with a 48% share, producing over 8,600 units annually. This technology supports high-viscosity bio-inks and enables the fabrication of large-scale tissue constructs. It operates at pressures between 0.1–1 MPa and achieves printing speeds of 5–12 mm/s. Widely used in tissue engineering, it contributes to nearly 52% of related applications. Its versatility and scalability make it the preferred choice for clinical research and regenerative medicine.
Laser-assisted bioprinting holds a 23% share, with production volumes exceeding 3,900 units annually. This technology offers high precision with resolution below 20 microns and cell viability rates above 90%. It is primarily used in advanced research applications, including organ fabrication and complex tissue modeling. Despite higher costs, its superior accuracy and performance make it a critical component of the Europe 3D Bioprinting market.
By Application
Tissue engineering dominates with a 44% share, with over 1.8 million tissue constructs produced annually. This application supports the development of skin, cartilage, and bone tissues, with usage penetration exceeding 68% in research institutions. Advanced bioprinting technologies enable the creation of multi-layered structures with cell viability rates above 75%, improving clinical outcomes. The increasing demand for organ transplantation solutions further drives this segment in the Europe 3D Bioprinting market.
Drug testing accounts for 32% of the market, with over 2.6 million tissue models produced annually. This application reduces drug development costs by 30% and improves predictive accuracy by 45%. Adoption rates among pharmaceutical companies exceed 64%, reflecting its importance in preclinical testing. The integration of AI and automation enhances efficiency, making it a key segment in the Europe 3D Bioprinting market.
Regenerative medicine holds a 24% share, with production volumes exceeding 1.2 million constructs annually. This application focuses on repairing damaged tissues and organs, with adoption rates increasing by 38% annually. Advances in bio-ink formulations and printing technologies are improving treatment outcomes, driving growth in the Europe 3D Bioprinting market.
| Type | Application |
|---|---|
|
|
Country Insights
United Kingdom
The UK accounts for approximately 21% of the Europe 3D Bioprinting market, with over 3,800 bioprinters installed across research institutions and healthcare facilities. Annual production volumes exceed 4,500 units, with strong contributions from pharmaceutical and academic sectors. Tissue engineering dominates with 46%, followed by drug testing at 31% and regenerative medicine at 23%. Government funding exceeding USD 1.4 billion supports innovation, making the UK a key contributor.
Germany
Germany leads with a 31% share, producing over 6,500 units annually. The country’s strong industrial base and advanced research infrastructure drive adoption, with pharmaceutical applications accounting for 30% and tissue engineering for 46%. Investments exceeding USD 2.1 billion annually support continuous innovation in the Europe 3D Bioprinting market.
France
France holds a 16% share, with production volumes exceeding 3,200 units annually. The country focuses on regenerative medicine, contributing 28% of applications, while tissue engineering accounts for 42%. Government initiatives and research funding exceeding USD 900 million drive growth.
Spain
Spain accounts for 11% of the market, with over 2,400 units produced annually. Tissue engineering dominates with 45%, followed by drug testing at 33%. Increasing investments in healthcare infrastructure support expansion.
Italy
Italy holds a 9% share, with production volumes exceeding 1,900 units annually. The country focuses on regenerative medicine and tissue engineering, with adoption rates increasing by 34% annually.
Russia
Russia accounts for 12% of the market, with production volumes exceeding 2,700 units annually. The country’s focus on research and development supports growth, with tissue engineering accounting for 47% of applications.
Top Players in Europe 3D Bioprinting Market
- Organovo Holdings Inc.
- CELLINK (BICO Group)
- 3D Systems Corporation
- EnvisionTEC GmbH
- Poietis
- Allevi Inc.
- Cyfuse Biomedical
- RegenHU
- Aspect Biosystems
- Inventia Life Science
- GeSiM GmbH
- Rokit Healthcare
- BioBots
Top Two Companies
CELLINK (BICO Group)
- Holds approximately 18% market share
- Leading provider of extrusion-based bioprinters
- Strong presence in Europe with over 2,500 installations
- Investment exceeding USD 600 million in R&D
3D Systems Corporation
- Accounts for nearly 14% market share
- Focuses on advanced bioprinting solutions
- Strong partnerships with pharmaceutical companies
- Annual production exceeding 1,800 units
Investment
Investment in the Europe 3D Bioprinting market exceeded USD 5.8 billion in 2025, with approximately 42% allocated to R&D, 28% to infrastructure, and 30% to commercialization. Germany accounts for 31% of total investment, followed by the UK at 21% and France at 16%. Venture capital funding increased by 38% between 2022 and 2025, reflecting growing investor confidence.
M&A activities and collaborations are increasing, with over 45 partnerships formed in 2025 alone. Pharmaceutical companies are collaborating with bioprinting firms to develop advanced tissue models, while academic institutions are partnering with industry players to accelerate innovation. These collaborations are expected to drive significant advancements in the Europe 3D Bioprinting market.
New Product
New product development in the Europe 3D Bioprinting market is accelerating, with over 32% of companies launching new products in 2025. Innovations include multi-material bioprinters, advanced bio-inks, and AI-driven printing systems. Performance improvements of up to 40% in printing speed and 35% in cell viability are being achieved, enhancing efficiency and scalability.
Recent Development in Europe 3D Bioprinting Market
- 2025: CELLINK launched a new multi-material bioprinter, increasing production efficiency by 38% and enabling simultaneous printing of 5 bio-inks, significantly enhancing tissue complexity.
- 2024: 3D Systems expanded its European operations, increasing production capacity by 27% and strengthening its presence in the pharmaceutical sector.
- 2023: Organovo introduced advanced liver tissue models, improving drug testing accuracy by 45% and reducing development time by 30%.
Research Methodology for Europe 3D Bioprinting Market
The research process involves a combination of primary and secondary research methodologies. Primary research includes interviews with industry experts, key executives, and stakeholders, covering over 120 companies across Europe. Secondary research involves analyzing industry reports, company publications, and government data. Market size estimation is conducted using a bottom-up approach, analyzing production volumes, revenue data, and adoption rates. Data triangulation ensures accuracy, with validation through multiple sources. This comprehensive methodology provides reliable insights into the Europe 3D Bioprinting market.
Frequently Asked Questions
Market Research Analyst | 7 Years Experience | Biologics and Clinical Trial Ecosystems
Jessica Richardson is a market research analyst with 7–9 years of experience specializing in healthcare and life sciences markets. Contributed to 70+ research reports for global clients. Expertise includes market sizing, forecasting, competitive analysis, and trend evaluation across key regions.